Demand for polyolefins in USA medical devices is set to grow at a 5.4% CAGR through 2036, driven by strict FDA quality regulations for device materials.
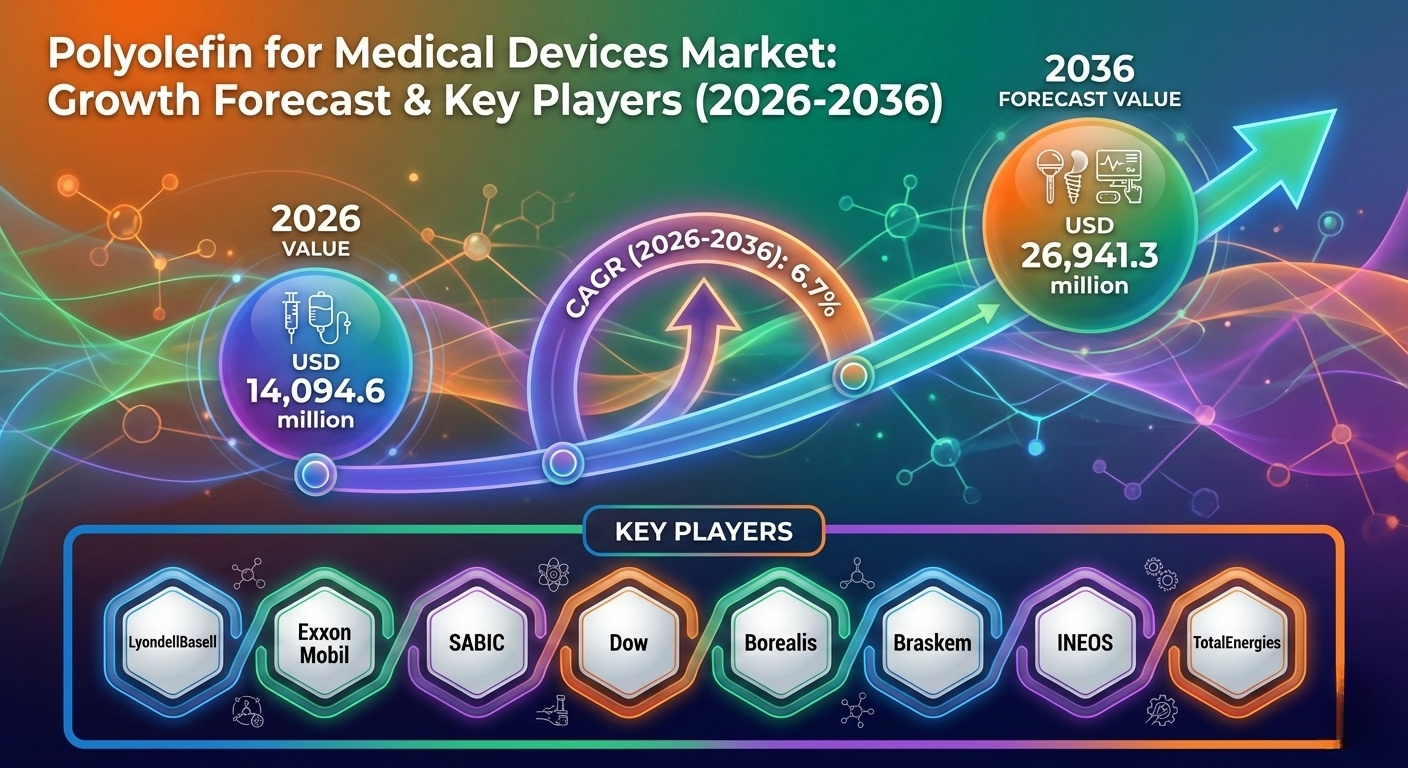
NEWARK, DE, UNITED STATES, February 26, 2026 /EINPresswire.com/ — The global polyolefin for medical devices market is entering a phase of sustained expansion, supported by rising demand for disposable healthcare products, advanced drug delivery systems, and cost-efficient medical-grade materials. According to the latest analysis by Future Market Insights (FMI), the market was valued at USD 13,174.2 million in 2025 and is projected to reach USD 14,094.6 million in 2026, ultimately growing to USD 26,941.3 million by 2036, reflecting a compound annual growth rate (CAGR) of 6.7%.
The market is expected to generate an absolute dollar opportunity of USD 12,846.7 million between 2026 and 2036, underscoring strong structural demand across healthcare systems globally. Growth is being driven by increasing adoption of disposable medical devices, expanding home healthcare, and the need for materials that meet strict regulatory and sterilization requirements.
Request Your Sample Now – Unlock Growth Potential and Discover Key Market Opportunities!
https://www.futuremarketinsights.com/reports/sample/rep-gb-32095
Market Overview: What is Driving Growth?
Polyolefins including polyethylene (PE), polypropylene (PP), ethylene vinyl acetate (EVA), and specialty olefin-based polymers, play a critical role in medical device manufacturing. These materials are widely used due to their biocompatibility, chemical resistance, flexibility, durability, and compatibility with sterilization processes.
They are essential in the production of syringes, IV bags, blood bags, catheter components, diagnostic casings, inhaler housings, insulin pens, and wearable drug delivery systems. As healthcare systems continue to prioritize infection control and patient safety, demand for these materials is accelerating.
A structural shift toward single-use and disposable medical devices is reinforcing long-term market growth. Disposable systems reduce cross-contamination risks and align with global infection prevention protocols, particularly in hospitals and clinical settings.
Key Market Drivers and Industry Trends
Several converging trends are shaping the polyolefin for medical devices market:
• Rising demand for disposable medical devices: Increased usage of syringes, IV systems, and diagnostic consumables is driving high-volume polymer consumption.
• Expansion of drug delivery technologies: Innovations such as auto-injectors, wearable insulin systems, and transdermal devices are increasing demand for high-purity materials.
• Regulatory emphasis on material safety: Stringent requirements related to biocompatibility, traceability, and sterilization stability are encouraging adoption of medical-grade polyolefins.
• Growth in home healthcare: Increased use of wearable and home-use medical devices is creating new application areas for advanced polymer formulations.
• Material innovation: Development of metallocene polypropylene (mPP), cyclo-olefin copolymers (COC/COP), and impact-modified polyolefins is enabling improved performance, clarity, and durability.
Despite these growth drivers, the market faces challenges from raw material price volatility, regulatory compliance costs, and competition from specialty engineering polymers, which can influence pricing flexibility.
Segment Analysis: Polyethylene and OEMs Lead Demand
By Polyolefin Type
Polyethylene (PE) is expected to dominate the market, accounting for 57.8% of total consumption in 2026. Its widespread adoption is attributed to:
• High biocompatibility and chemical resistance
• Reliable performance under gamma and ethylene oxide sterilization
• Ease of processing for high-volume manufacturing
• Compatibility with medical tubing, IV bags, and packaging films
Polypropylene (PP) also maintains strong adoption due to its strength, clarity, and sterilization versatility, making it suitable for syringes, diagnostic housings, and rigid components.
By End User
Original Equipment Manufacturers (OEMs) are projected to hold 42.8% of market share in 2026, driven by their central role in large-scale production of medical devices. Polyolefins support OEM priorities such as:
• Regulatory compliance and traceability
• Lightweight design and cost efficiency
• Scalability for high-volume disposable products
• Customization for minimally invasive and wearable technologies
Regional Outlook: Asia Pacific Leads Growth Momentum
The global market exhibits varied growth patterns across regions:
• China (7.2% CAGR): Fastest-growing market, supported by expanding healthcare infrastructure, increased medical device manufacturing, and export capacity.
• India (6.8% CAGR): Growth driven by domestic manufacturing expansion and government initiatives such as the Production Linked Incentive scheme.
• Brazil (6.1% CAGR): Rising demand for diagnostic equipment and healthcare modernization supports polymer consumption.
• United States (5.4% CAGR): A mature market characterized by innovation, strict regulatory oversight, and demand for high-purity materials.
• Germany (5.1% CAGR): Growth driven by regulatory compliance under the EU Medical Device Regulation and sustainability initiatives.
Asia Pacific remains the fastest-growing production hub, benefiting from increased hospital infrastructure and domestic manufacturing capacity.
Competitive Landscape: Focus on Compliance and Supply Reliability
The polyolefin for medical devices market is moderately concentrated, with major global players including:
• LyondellBasell Industries N.V.
• Exxon Mobil Corporation
• Saudi Basic Industries Corporation (SABIC)
• The Dow Chemical Company
• Borealis GmbH
• Braskem S.A.
• INEOS Group Holdings S.A.
• TotalEnergies SE
• Reliance Industries Limited
• Chevron Phillips Chemical Company LLC
Competition is primarily based on material purity, regulatory compliance, supply reliability, and long-term contracts with medical device manufacturers. Companies with integrated feedstock access and global production networks maintain a competitive advantage.
Strategic Takeaways for Industry Stakeholders
• Invest in high-purity, sterilization-compatible polymer grades to meet evolving healthcare regulations
• Strengthen partnerships with drug delivery and wearable device innovators
• Expand regional production capacity to mitigate supply chain risks
• Focus on traceability and compliance frameworks to maintain regulatory approval
• Explore sustainable and lower-carbon polyolefin solutions for future healthcare applications
Why FMI: https://www.futuremarketinsights.com/why-fmi
Have a Look at Related Research Reports on the Packaging Domain:
Spout Pouch Market https://www.futuremarketinsights.com/reports/spout-pouches-market
Medical Device Packaging Market https://www.futuremarketinsights.com/reports/medical-device-packaging-market
Metal Packaging Market https://www.futuremarketinsights.com/reports/metal-packaging-market
Specialty Tin Cans Market https://www.futuremarketinsights.com/reports/specialty-tin-cans-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()